Your Next Generation Cell & Gene Therapy Development Partner
The fast-paced Cell & Gene Therapy landscape brings the potential for health improvements for patients waiting for medication and disease treatments. Cell & Gene therapy biotech and pharma communities face the challenge of bringing novel therapeutics to the market, within tight deadlines. Meeting the demands of cell & gene therapy development at every stage of the drug development process requires a strategic partner capable of dealing with highly complex clinical trial designs. To deal with this complexity, the industry needs highly customized laboratory solutions.

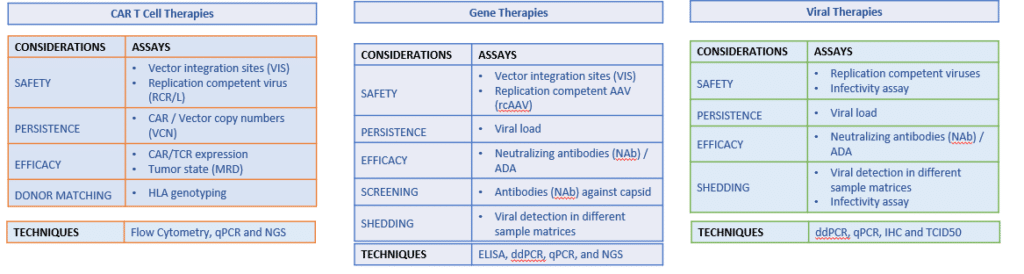
As an industry leader in tailored solutions, Cerba Research offers a full suite of services for Cell & Gene Therapy clinical trials, from asset identification, working through the non-clinical studies, and transitioning into clinical trials, from dose study applications to expansion and long-term follow-up. Tailor-made solutions are offered for CAR-T, Gene, and Cell Therapy trials.
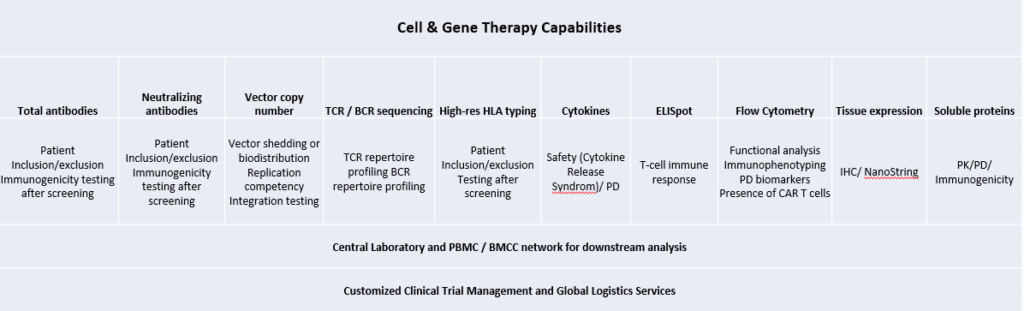
We offer customized assay design and fit-for-purpose validation. Cerba Research works with the biotech and pharma communities as a fully integrated partner, using its knowledge and scientific expertise to help sponsors shape testing protocols, patient cohorts and identify the right testing strategy to secure successful outcomes. Upon initial validation in the EU or US region, assays can be transferred and implemented around the globe, according to the need of the trial. Cerba Research offers a full suite of immunology, genomics and flow cytometry assay solutions
Central Laboratory & Clinical Trial Logistics for Cell & Gene Therapy Development
Our unique combined specialty laboratory and central lab services offer the best of both worlds, allowing our customers to have one partner for all test services. From choosing the most appropriate geographies and labs to our best-in-class logistic network, securing the integrity of samples. Cerba Research’s fully integrated specialty logistic portfolio includes the following:
•Accessibility to a global network of laboratories capable of complex specialty testing for cell & gene therapy development.
•Global PBMC/ BMCC network – secure sample integrity for further downstream analysis.
•Samples are monitored 24/7, temperature-controlled, and transported to reach their destination on time.
•Kit manufacturing, assembly and worldwide distribution to support Cell & Gene Therapy Clinical Trials.
Cerba research is the go-to partner for tailored logistic solutions for the next generation of cell and gene therapies. Here you can read more about our capabilities to set up and operationalize a highly customized gene therapy targeting Spinal Muscular Atrophy (SMA).
Webinars – Engage with our scientific communities for the latest insights in C&G therapy development
Relevant Content

Reach out to our experts to see how we can help you advance your research and development in rare diseases
Contact Us
