Our specialty laboratory solutions can fit into your current clinical development plans. Even if you are using another central laboratory provider, our agile approach and customer-centric view allows you to communicate scientist to scientist easily.
Services

Pre-clinical services
As a leading diagnostic and clinical trial operation service company specializing in virology and immunology, Cerba Research provides a wide range of preclinical services to test the safety, effectiveness, PK, and (immune) toxicology of antivirals, drugs, antibodies, and vaccines that target infectious diseases.
Virology and Vaccine Lab
With our services, we assist the pharmaceutical, biotech and academic communities from early preclinical research, including in vivo models, through large-scale, late-stage clinical trial programs for vaccines, monoclonal antibodies, and antivirals

Assay Development and Technology Transfer
Cerba Research also develops tailor-made virological, immunological and molecular assays and is highly experienced in all phases of assay development.

Immunoassays
You can rely on Cerba Research to outsource your research questions, including efficacy, safety, passive immunization, immunogenicity, pharmaco kinetics, pathogenicity, antiviral therapy, antiviral prophylaxis, drug delivery, resistance mutants, and more.
Flow Cytometry
Connect with our scientific team to learn how FCM can enhance your research and help you develop the panels you need.
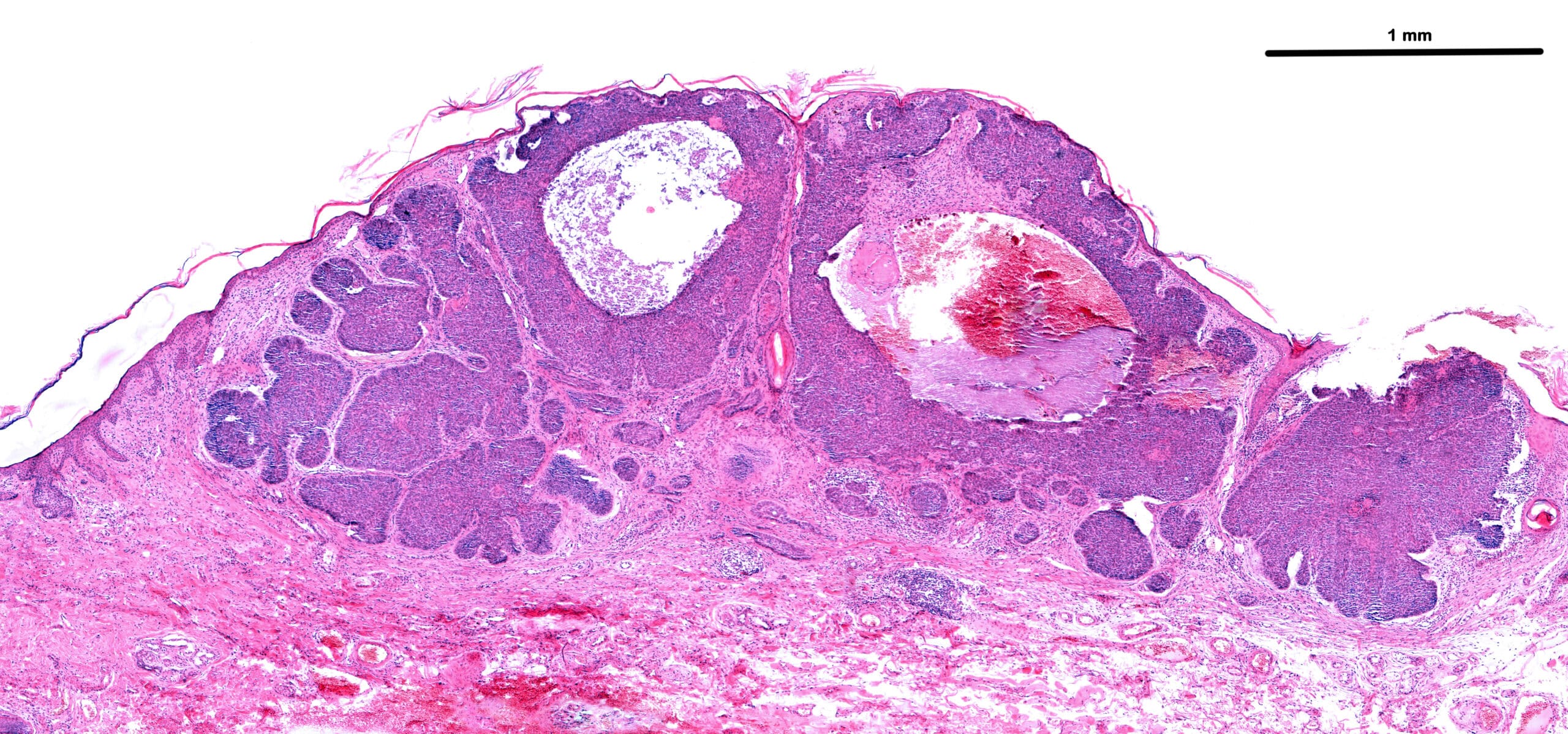
Anatomic Pathology
We’re committed to delivering gold-standard tissue sourcing, processing and analysis services.
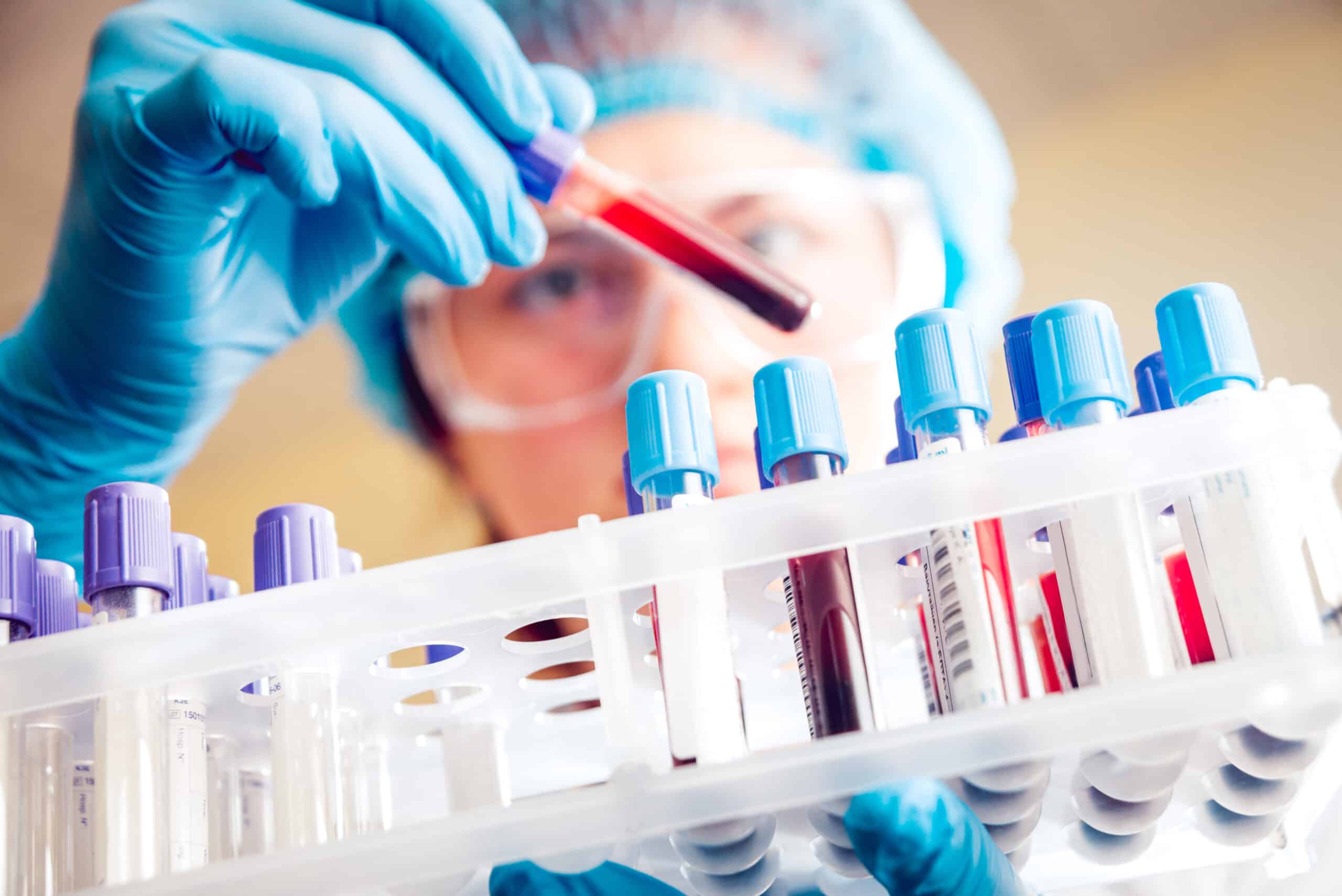
Pre-analytical/ PBMC Processing
At Cerba Research we are specialized on functional and analytical tests downstream of PBMCs isolation. We guarantee customers a high level of quality from sample collection to processing, shipping, handling, storage and analysis worldwide.
Data Intelligence/ Bioinformatics
Cerba Research has developed unique pipelines specifically for the analysis of viral and microbiome NGS data. Our team of experienced scientists and bioinformaticians work together to provide fully GxP and FDA compliant pipelines that have been scientifically validated. We can help customers to set up data transfer formats following CDISC standards.